Mapping The Mutational Landscape
Our protein biochemistry capabilities include advanced selection methodologies in both yeast and mammalian cell systems. These tools can interrogate large numbers of mutations in cell surface receptors and globular proteins, rapidly identifying the impact of amino acid changes and offering unique guidance for protein optimization. Performing this in a human cell-based system also means high quality, and highly physiologically-relevant data.
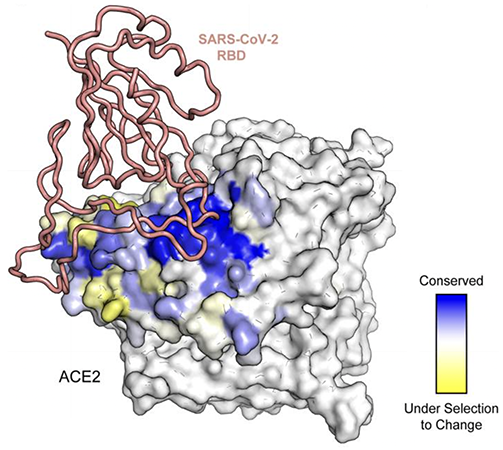
Exhaustive single point mutations of ACE2 were tested by yeast display to identify mutations that affect binding to SARS-CoV-2’s Spike protein. By sequencing the mutations that retained binding, we can explore sequence diversity. Deep mutational scans identified conserved positions (in blue) that are critical for binding. Positions with high mutation tolerance (in yellow) are under selection to change. From “Narayanan KK, Procko E. Deep Mutational Scanning of Viral Glycoproteins and Their Host Receptors. Front Mol Biosci. 2021 Apr 9;8:636660.”
Eric Procko – Director of Discovery at Cyrus
Mutational Scanning
INTERESTED IN
PARTNERING WITH US ON
PROTEIN TERAPEUTICS?